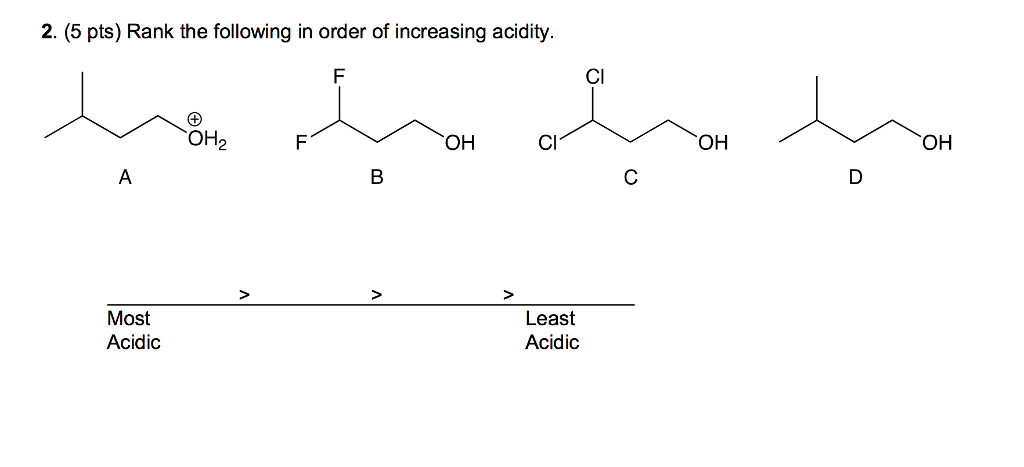
D Which of the following is a Lewis base?Į: None of the above are Lewis bases. 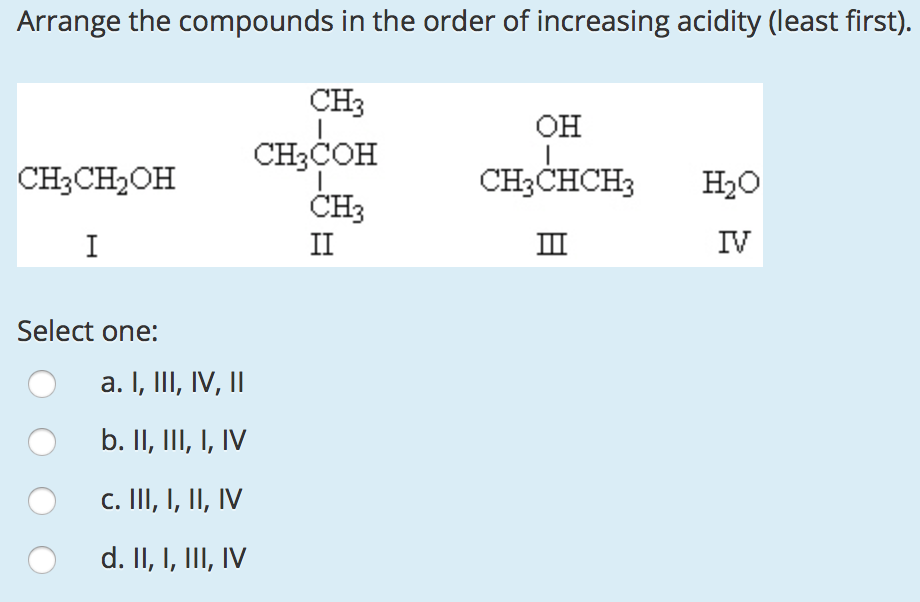
electron pair donor Lewis base _ B consider the two lewis structures given below which of the following statements is correct?Ī: In a reaction, structure 1 would function as a Lewis base while structure 2 would function as a Lewis acid.ī: Structure 2 can be classified as both a Brønsted-Lowry and a Lewis base.Ĭ: Structure 1 can be classified as both a Brønsted-Lowry and a Lewis acid.ĭ: Structure 2 would have similar Lewis acid-base properties as those of AlCl3. Identify the strongest conjugate base.ĭ: CHO2- D Which ion will be basic in aqueous solution? Produces protons in an aqueous solution Arrhenius acid _ produces hydroxide ions in aqueous solution Arrhenius base _ proton donor Brønsted-Lowry acid _ proton acceptor electron pair acceptor C Which of the following is a Lewis acid?Į: None of the above are Lewis acids. 
CH3NH3NO3, BaBr2, NaNO2 Classify each salt as acidic, basic, or neutralīasic: A Consider the given acid ionization constants.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
